Your “MTHFR” Is Just a Riboflavin Deficiency
Or one of hundreds of other genetic impairments in energy metabolism.
Deficient methylation can lead to fatty liver disease, birth defects, cardiovascular disease, fatigue, poor exercise capacity, histamine intolerance, difficulty ignoring negative thoughts and thought patterns, depression, anxiety, obsessive compulsive disorder, histamine intolerance, and possibly cancer.
MTHFR is an important enzyme involved in methylation that is needed to support protection against all these things. Further, low MTHFR activity leads to loss of the amino acid glycine, which could hurt blood sugar, sleep, joint health, risk of psychosis, and make you startled more often or increase muscle tension.
There are two common MTHFR genetic variations, C677T and A1298C that hurt MTHFR activity. These are so common that you probably have at least one of them.
My MTHFR Protocol is designed to cover all the nutritional bases impacted by these polymorphisms, and many people swear by it. For example, Beth C wrote this:
I'm 42, and have an 81% decrease on my methylfolate score. I started this protocol a few weeks ago, and:
* my RHR is down to high 40s-mid 50s from the mid-60s;
* I haven't had a brain fog episode once -- very exciting to be able to do my business books whenever I have time, and not during a mysterious window of opportunity;
*HRV is is trending up;
*No more bouts of dyspnea;
*Energy and exercise tolerance has greatly improved;
*100% O2 sat for the first time in about two years. I live at altitude.
I feel like I have my life back. Thanks, Chris.
On the flip side of this, alishabd wrote on my Instagram that methylation “can be impossible to fix. 😢”
The reality is MTHFR should usually be so easy to fix with a few dietary modifications that my protocol is completely unneeded. The 133 comments on Start Here for MTHFR and Methylation, however, would seem to argue otherwise.
Why the discrepancy?
Because if “MTHFR” is difficult, let a lone impossible, to fix, then it almost certainly involves a stealth contribution from any one of more than two dozen nutrient deficiencies or hundreds of genetic impairments in energy metabolism.
The solutions to this are some biochemical detective work using comprehensive nutritional screening and comprehensive screening for energy metabolism.
In fact, if your health is worse than that of every fourth person around you, it almost certainly is driven by something beyond one of these common MTHFR polymorphisms, because one in four people are hit by the worst of them.
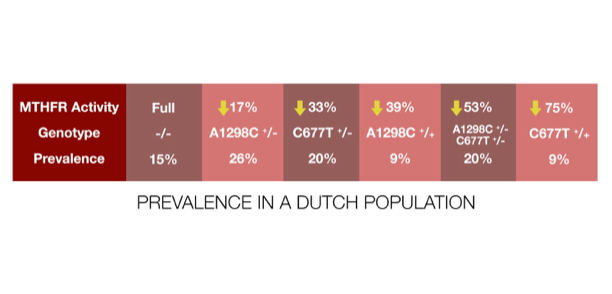
Here is the prevalence in a Dutch population:
Only 15% have the fully functional enzyme, and the various combinations of common polymorphisms lead to a nearly even, graded distribution of MTHFR activity from high to low across the other 85%. Even the worst combination, homozygous for C677T, is found in 25% of people.
At first glance such an even distribution of MTHFR activity suggests a spectrum on which a tradeoff occurs. That would mean there are costs and benefits to high and low activity, so variation is preserved in the population.
However, as covered in Banish Your Headaches, Fatigue, and High Blood Pressure With Riboflavin, 1.6 milligrams of riboflavin a day above and beyond average dietary intakes normalizes homocysteine in individuals with homozygous C677T. Baseline intakes of riboflavin are around 1.5 milligrams per day on modern diets, so this implies that C677T achieves normal function on a dietary intake of about 3 milligrams of riboflavin per day. This would be a achieved on a diet rich in liver, kidney, and heart (or lots of almonds, at the expense of the oxalate). Thus, the likely explanation of why these mutations are so common is that our ancestors ate nose-to-tail, got twice as much riboflavin as we did, and these polymorphisms did not hurt them at all in that context.
Thus, there is no reason to suspect a tradeoff. Rather, there is reason to think these polymorphisms proliferated because they didn’t matter.
Experimental models suggest that C677T makes the MTHFR enzyme more sensitive to heat-induced loss of riboflavin binding. Body heat is sufficient to stress the enzyme enough to cause that loss. The position of C677T and A1298C are very far from one another in both the gene and the enzyme, and there is no reason to believe that riboflavin would normalize the function of A1298C. Indeed, there are over 200 other much more severe much more rare mutations in MTHFR that have been identified, and only 5 of them respond to riboflavin because the others affect positions of the enzyme unrelated to riboflavin binding. Thus, riboflavin is not a cure-all for all things MTHFR.
However, the A1298C mutation does not raise homocysteine reliably unless it is combined with one copy of C677T, and this suggests that its role in decreasing MTHFR activity is just too minor to have a major effect that rises above the statistical noise caused by the many other factors that cause variation in homocysteine.
As I pointed out in How to Interpret the Genova Methylation Panel, homocysteine is not the be-all, end-all of methylation. You can have normal homocysteine but have your methylation sapped in other ways. However, those other ways are not directly related to MTHFR. Elevated homocysteine is a reliable metric of low MTHFR activity, so if A1298C cannot prove itself to be an independent factor in homocysteine, that means its own impact on MTHFR activity without any help from C677T is drowned out by the noise of the many other factors impacting methylation. That doesn’t make it meaningless, but it does mean it shouldn’t get particularly special attention in the context of methylation.
So, ancestrally, these polymorphisms didn’t matter, because C677T needs to get hit to make A1298C rise above the noise, and C677T doesn’t get hit on ancestral intakes of riboflavin.
Yet, anecdotally, people all over the internet swear that their MTHFR is the bane of their health. As alishabd lamented above, some swear such problems are unsolvable. This perception is most likely the result of a failure to realize that MTHFR is incidental to difficult-to-solve “MTHFR” problems and the missing link to the solution is a stealth contribution from deficiencies of any one of over two dozen nutrients or hundreds of other genetic impairments in energy metabolism.
There are at least 26 nutrients involved, so assessing the role of nutrients in methylation is a task for the Comprehensive Nutritional Screening.
Consider these:
MTHFR’s job is to use riboflavin (in the form of FAD) to take electrons from niacin (in the form of NADPH) to add them to what becomes the methyl group of methylfolate, which then gets passed on to vitamin B12, which then passes it to homocysteine to make methionine.
The NADPH in this reaction comes from glucose in the pentose phosphate pathway. This requires the enzyme lactonase, which requires magnesium or manganese and zinc. It also requires the enzyme transketolase, which requires thiamin.
The carbon atom that makes up the methyl group of methylfolate needs to be present before MTHFR does its thing, and this typically comes from serine or glycine. Both of these amino acids are found in dietary protein, though on a mixed carbohydrate-inclusive diet, they are primarily synthesized from glucose. The synthesis of serine from glucose requires the enzyme phosphoserine aminotransferase, which requires vitamin B6.
That carbon atom can also come from excess methionine that was buffered by methylating glycine and later harvested from methylated glycine metabolites in the mitochondria to yield formate. This requires the enzyme glycine N-methyltransferase, whose expression is increased by vitamin A; sarcosine dehydrogenase and dimethylglycine dehydrogenase, which require iron; and mitochondrial formyltetrahydrofolate synthetase, which requires potassium.
The carbon atom can also be gotten from glycine using the glycine cleavage system, which adds lipoic acid and vitamin B5 (as coenzyme A) as cofactors.
If formate is used to supply the carbon atom of the methyl group, MTHFD1 must use ATP to make it happen. This opens the door to the full suite of nutrients needed for ATP synthesis, which adds biotin (for pyruvate carboxylase to run the citric acid cycle), copper (for complex IV of the mitochondrial respiratory chain), iodine (to make thyroid hormone, which raises the rate of ATP synthesis), sulfur (for the iron-sulfur complexes of the respiratory chain), sodium (for the transport of many of these components).
The amino acid methionine from dietary protein is always the starting point to provide methyl groups, and MTHFR’s role is to recycle it after it becomes homocysteine. You can never get the “perfect” amount of methionine to keep this cycle running without a little bit of excess, and that excess will always require molybdenum for its clearance.
Oxidative stress will lead to excessive breakdown of homocysteine and make it unavailable to be recycled by MTHFR. Antioxidant protection adds vitamin C, vitamin E, and selenium to our list.
In addition, MTHFR activity will need to be higher if choline and betaine run low and therefore cannot be used to support methylation through the alternative enzyme betaine homocysteine methyltransferase.
This alone is a list of 26 nutrients.
The role of ATP opens up the door to hundreds of genetic impairments in energy metabolism. In How I Found My Health “Super Unlock” After 20 Years of Research and 20,000 Genes Tested, I estimated that each person has one to six nutritionally actionable genetic mutations in energy metabolism that are orders of magnitude more important than the other 14 million sites of genetic variation found in everyone. This means that genuinely drilling down into the stealth contribution to deficient MTHFR activity means running comprehensive screening for energy metabolism.
The energy metabolism screening can reveal needs for nutrients that the nutritional screening will not reveal. For example, you could have optimal riboflavin status due to a nose-to-tail diet, but you could have a riboflavin-responsive impairment in an enzyme such as ACAD9 that will sap your ATP production unless riboflavin is brought to supraphysiological levels with doses such as 75 milligrams or 400 milligrams per day. The low ATP would hurt MTHFR activity regardless of the presence of MTHFR polymorphisms.
Thus, someone could be homozygous for C667T and also have the ACAD9 mutation. At 1.5 milligrams per day of riboflavin, the comprehensive nutritional screening shows that their riboflavin status is fine, but they have a 75% genetic hit against their MTHFR. At 3 milligrams per day of riboflavin, their C677T homozygosity becomes completely irrelevant, but their MTHFR activity is indirectly sapped by low ATP. At 75 milligrams per day of riboflavin their ACAD9 is optimized, their ATP levels improve, and MTHFR functions normally.
This is just one of hundreds of possible reasons for low ATP, however, thus the importance of the screening, which could reveal hundreds of other such solutions.
So:
The low-hanging fruit of MTHFR is to get dietary riboflavin to 3 milligrams per day. Track your diet if needed.
Right above that: my MTHFR Protocol.
Next-level: fix any nutrients found in the comprehensive nutritional screening.
Highest-level: fix any problems found in the comprehensive screening for energy metabolism.
Methylation is never unfixable, but it will often require thinking outside the homocysteine/folate/B12 box.


Hi Chris, love your work, your unique approach gave me insights for my work.
I have a HUGE question for you: You say that Riboflavin 5'-Phosphate is worse than cheapo staff, because our gut is cutting of that 5-phosphate of everything, but at the same time you recommend Pyridoxal 5'-phosphate as a better form of B6. What am I missing?
Is liverwurst a good enough source of riboflavin?