The Sweet Truth About Liver and Egg Yolks — Choline Matters More to Fatty Liver Than Sugar, Alcohol, or Fat
In a recent post, I pointed out that perhaps as many as 100 million Americans have some degree of fatty liver disease. Why? Alcohol has been blamed since the 1800s, but we currently have an epidemic of nonalcoholic fatty liver. Some researchers, such as Dr. Robert Lustig, are making the case against fructose. Naturally, the nutritional establishment and the media will blame anything on saturated fat, and fatty liver is no exception, even when the “saturated fat” is corn oil.
After studying the relevant literature and tracing it much further back in time than anyone else ever bothers to, I've come to the conclusion that neither fat nor sugar nor booze are the master criminals here. Rather, these mischeivous dudes are just the lackeys of the head honcho, choline deficiency. That's right, folks, it's the disappearance of liver and egg yolks from the American diet that takes most of the blame.
More specifically, I currently believe that dietary fat, whether saturated or unsaturated, and anything that the liver likes to turn into fat, like fructose and ethanol, will promote the accumulation of fat as long as we don't get enough choline. Once that fat accumulates, the critical factor igniting an inflammatory fire to this fat is the consumption of too much PUFA (polyunsaturated fat from vegetable and perhaps fish oils).
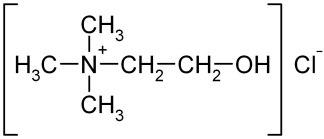
Most reviews about nonalcoholic fatty liver disease (NAFLD) trace the disease back to a 1980 paper published by Jurgen Ludwig and several of his colleagues from the Mayo Clinic (1). But Ludwig's group never claimed to have discovered NAFLD. On the contrary, they simply came up with the name nonalocholic steatohepatitis, which they abbreviated “NASH.” They took credit for achieving three things by inventing this new word (2). First, NASH is really easy to say. It only takes one syllable. Second, giving a convenient name for the disease encouraged an organized approach to researching it. Third, it stopped physicians from assuming their patients were lying about not consuming alcohol just because they had an inflamed liver.
As Ludwig's group acknolwedged, Samuel Zelman had produced the first case series of obesity-associated NAFLD patients back in 1952 (3). Zelman launched his investigation after observing fatty liver in a hospital aide who drank more than 20 bottles of Coca-Cola every day. This was before the days of the obesity epidemic, so it took him a full year and a half to find 20 obese people who weren't alcoholics. All but one of them had some indication of liver damage, and about half of them had pretty substantial NAFLD.
But fatty liver goes back even further than that. According to one paper I found, fatty liver had been identified in type 1 diabetes at least as far back as 1784 (4). By the 1930s, some physicians recognized fatty liver as a common occurrence in severe cases of diabetes and it tended to spontaneously resolve when they treated the patients with insulin and a low-carbohydrate diet that included 100 grams of bread and 100 grams of fruits and low-carbohydrate vegetables, with the remainder of energy requirements coming from fat (5).
Severe type 1 diabetes is quite a special case and it is likely that in these instances it was the severe metabolic derangement causing the fatty liver. This, however, quite clearly cannot explain why as many as 100 million Americans or more currently have the disease. Nevertheless, it was the study of type 1 diabetes animal models that led to the discovery of the critical role of choline in preventing and curing fatty liver.
Diabetes and Choline
Physiologists first identified the role of insulin deficiency in type 1 diabetes by studying the disease in dogs. In 1889 they produced diabetes by simply taking out the whole pancreas from these dogs and, after scrambling for a couple decades to identify the active component, they cured the diabetes with insulin in the early 1920s (6). As cured as their diabetes was, the insulin-treated dogs nevertheless developed severe fatty liver degeneration and ultimately died of liver failure. Adding raw pancreas to their diet, which was composed of lean meat and sucrose, cured the problem. As researchers attempted to discover what it was in raw pancreas that cured the disease, they found in the early 1930s that egg yolk lecithin, which is abundant in choline, could cure it (7). And then they found that choline alone could cure it (8).
It later turned out that the dogs became deficient in choline and methionine without a pancreas because they weren't producing the digestive enzymes needed to free up those nutrients from the foods they were eating. Thus, simply providing them with the digestive enzyme trypsin could cure the fatty liver (9).
We now know that choline is necessary to produce a phospholipid called phosphatidylcholine (PC). This is a critical component of the very low density lipoprotein (VLDL) particle, which we need to make in order to export fats from our livers. The amino acid methionine can act as a precursor to choline and can also be used to convert a different phospholipid called phosphatidylethanolamine directly into PC. Thus, the combined deficiency of choline and methionine will severely impair our abilities to package up the fats in our livers and to send them out into the bloodstream (10).
High-Fat Diets and Choline
In 1932 a group of researchers decided to replicate the fatty liver seen in depancreatized dogs in a nondiabetic rat model. What better way to stuff their livers with fat? Feed them fat! Seemed simple enough. And, in fact, it worked. Althogh they had trouble reproducing the fatty liver with different colonies of rats or during the summer heat, they produced fatty liver in certain colonies of rats during the winter by replacing 40% of their ordinary cereal-based diet with beef drippings. Lecithin derived from egg yolk or beef liver (11) or simply choline itself (12) cured the disease.
These experiments were back in the day, before researchers realized that methionine would be able to indirectly cure choline deficiency. Another group of researchers had the bright idea of trying to replicate this experiment in a group of rats who were consuming sufficient protein to maximize growth. So they fed them 40% beef drippings but replaced another 20% of their cereal grains with the milk protein casein.
Lo and behold, these researchers discovered a now well known phenomenon that occurs during the process of scientific discovery called the “epic fail” (13).
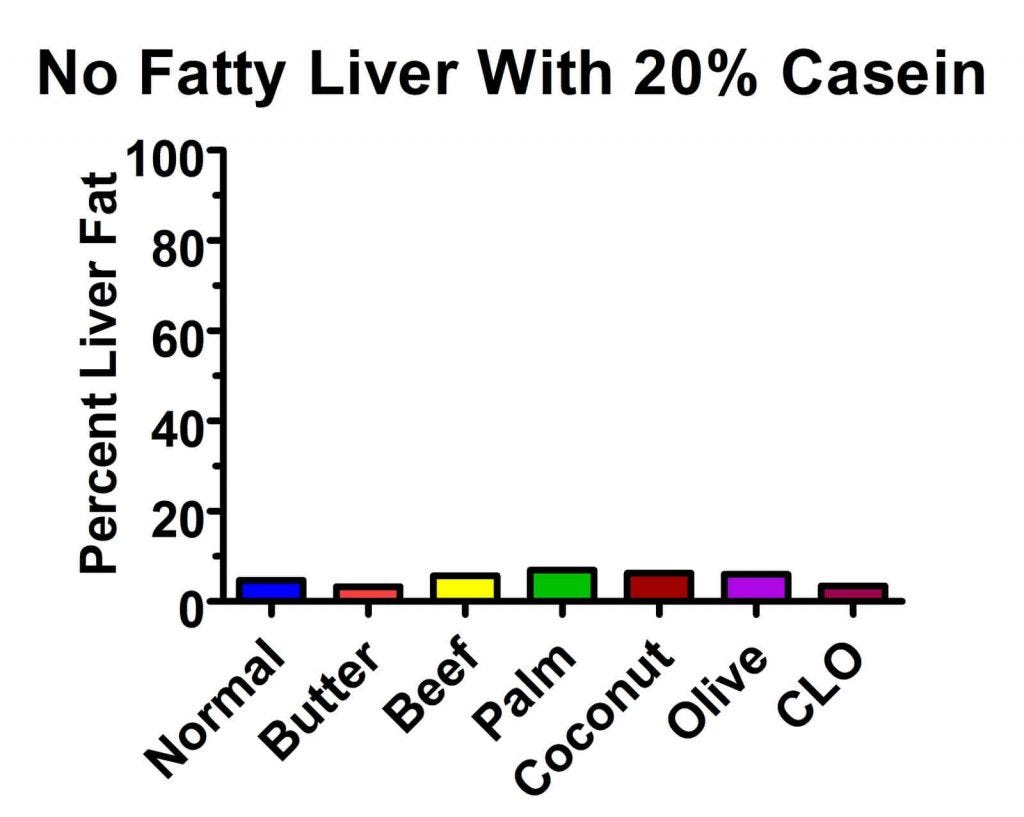
All of the groups had average levels of liver fat under 7% and could hardly be said to have gotten fatty liver of any sort.
So the researchers had an idea. Perhaps it was the casein they fed them that was the problem. And indeed, their hypothesis turned out to be correct: on a choline-free, 40% beef dripping diet, reducing the casein from 20% to 5% doubled the level of fat in the liver (14).
Those of you who have been reading my blog for the past few months may remember that in the wake of Denise Minger's shredding apart of the China Study's epidemiological data, I dug deep down into the buried secrets of Dr. T. Colin Campbell's rat research, and found that he attributed the “protective” effect of 5% casein diets to the massive fatty liver that the rats developed. As described in “The Curious Case of Campbell's Rats” and the associated addendum, the membrane containing a drug-detoxifying enzyme that he blamed for liver cancer got stuffed with 3-4 times more fat, which may have clogged up the enzyme and stopped it from working. Well, it turns out that others had shown a similar effect of such a diet on liver fat decades earlier.
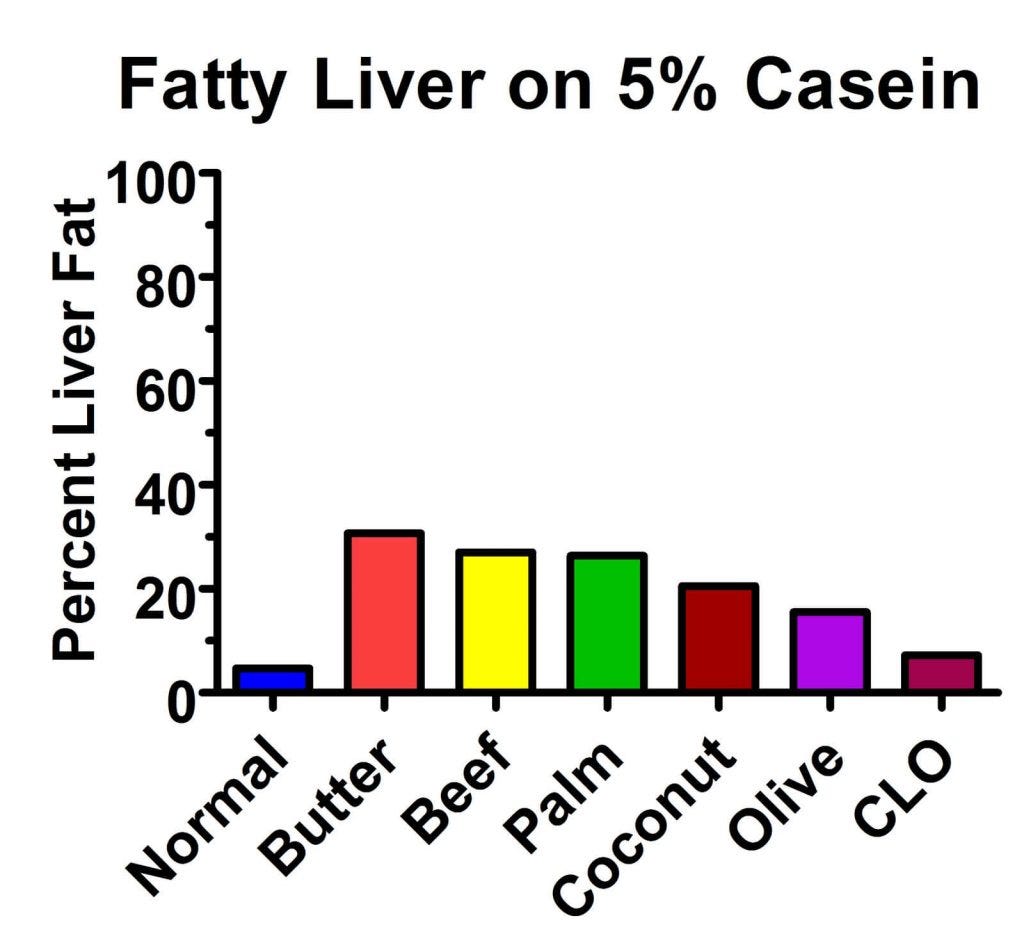
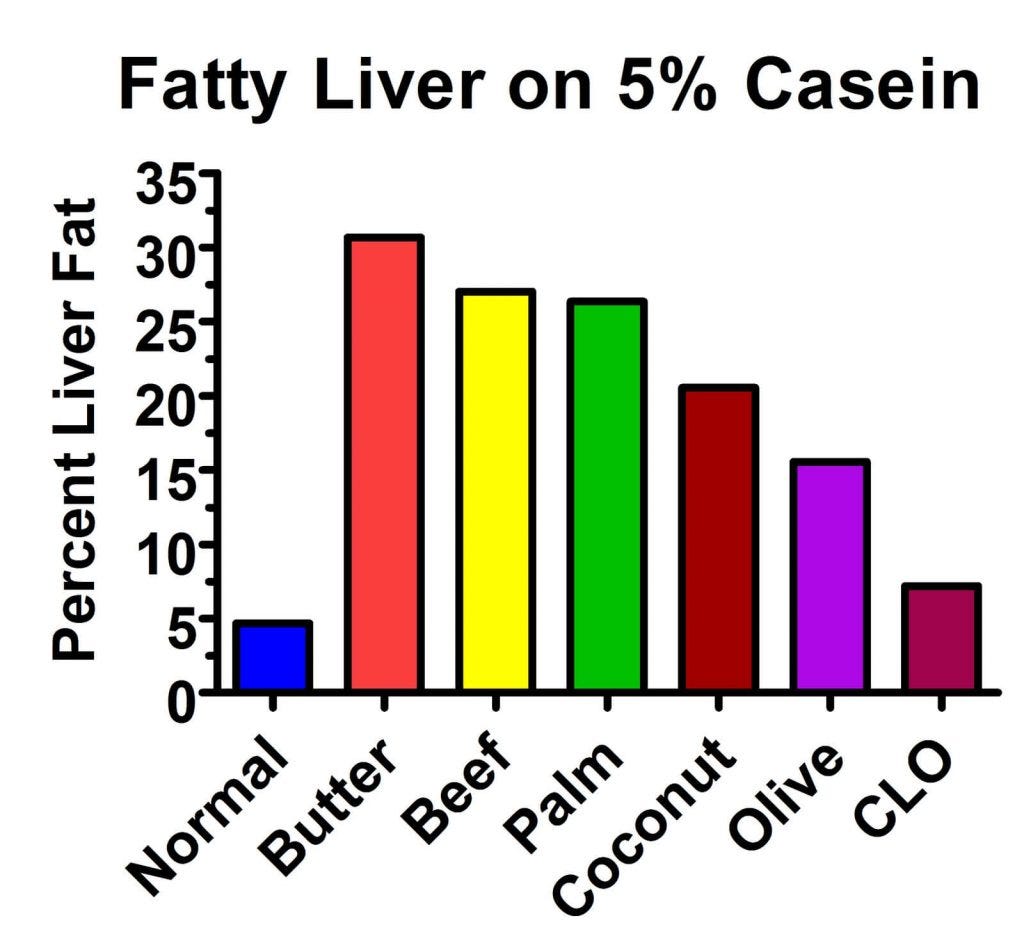
Back to the 1930s. So these researchers repeated their experiment with the different fats, this time using 5% casein. Ta-da! The experiment worked much better (13):
The butter produced the worst fat accumulation, where almost a third of the liver was fat! The animals taking cod liver oil tended to eat a bit less food than the others, which undoubtedly confounded the results at least to some degree, but it still seems from this graph that the more saturated the fat and the longer-chain the fat, the worse the fatty liver was. In fact, if we blow up the graph, we can see this more easily:
Whoa! Hold your horses, you might be thinking. Mr. Masterjohn, haven't you been telling us that saturated fat protects against fatty liver disease?
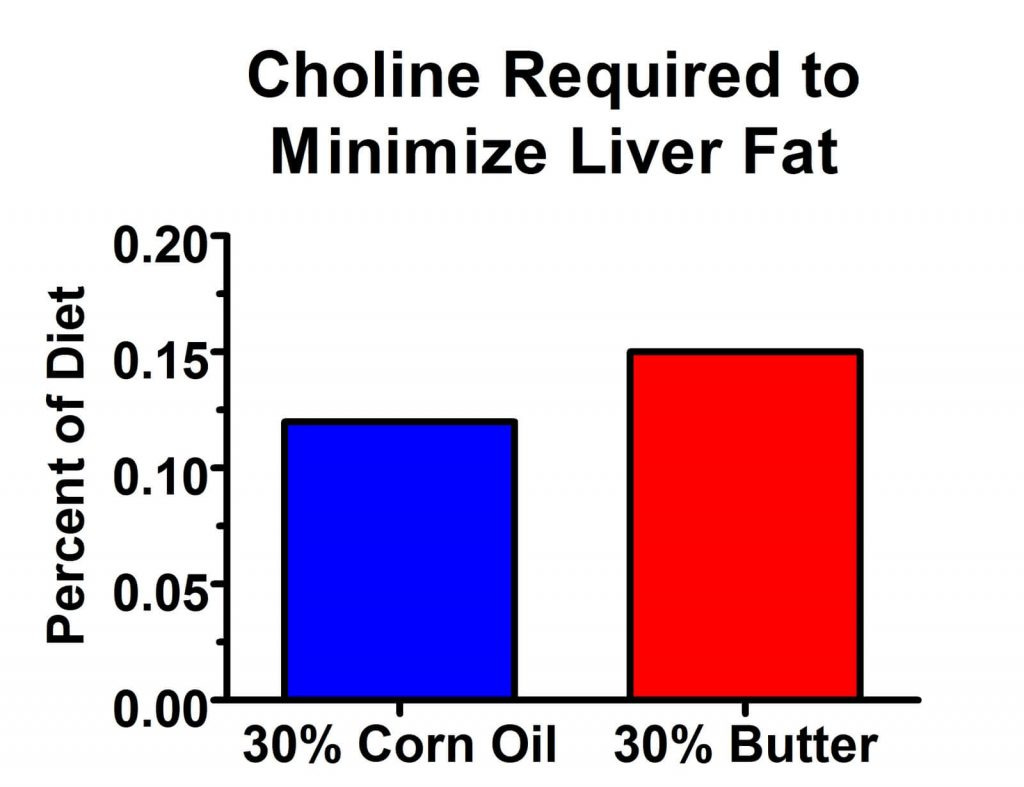
Well, yes I have, and I stand by that. But it appears that things are a little more nuanced than they first appeared. As it turns out, saturated fats increase the choline requirement a bit more than PUFAs do. Take a look at the results of this 1957 paper that tested the effect of butter and corn oil on the choline requirement (15):
As it turns out, the choline requirement is about 30% higher on a 30% butter diet than on a 30% corn oil diet. Why would this be? It's not entirely clear, but I have a good guess. As I pointed out in my PUFA Report, “How Essential Are the Essential Fatty Acids?”, studies in rats, humans, and other primates show that 18-carbon PUFA are burned for energy at an extraordinarily high rate compared to other fats. In rats, 60% are burned for energy, 20% are broken down into basic building blocks to make more saturated fatty acids, and most of the rest are secreted into the fur. They do accumulate over time at a slow rate, but the body seems to sense that these fatty acids are an unnecessary oxidative liability and tries to get rid of them however it can. Thus, perhaps saturated fats require more choline to get them out of the liver because they don't scare the liver into burning them for energy so quickly.
In any case, the clear picture that is emerging is that dietary fat increases the choline requirement, and that high-fat diets promote fatty liver only when the level of choline in the diet is insufficient to meet the extra demand for it caused by the increase in fat.
Fructose, Sucrose, Ethanol, and Choline
The buzz is all about fructose nowadays, but the role of fructose in fatty liver has been known since the 1930s, just like the role of fat. It also played a critical role in the modification of the AIN-76A standards for purified lab rat diets to the AIN-93 standards in 1993, when sucrose was switched to corn starch, like I explained in “They Did the Same Thing to the Lab Rats That They Did to Us.” Fructose can be provided in the diet as free fructose or as sucrose, which is half fructose and is the type of sugar found in refined, white table sugar. Fructose is different than glucose in that it goes straight to the liver rather than dispersing throughout the body; thus, the liver converts much of it to fat, sending the fat back into the bloodstream — providing it has enough choline. Ethanol, the type of alcohol we get drunk from, is metabolized similarly (16).
The first studies showing that fructose played a role in fatty liver disease developed out of the discovery of the essential fatty acids. I've recounted the history of this discovery in my recent article, “Precious Yet Perilous — Understanding the Essential Fatty Acids.” In 1924, George Burr joined the laboratory of Herbert Evans, where Evans and Katherine Scott Bishop had recently discovered vitamin E. Evans and Bishop were having trouble reproducing their vitamin E-deficient diet, and Burr helped them develop a highly purified diet based on reprecipitated casein and recrystallized sucrose. The new diet produced a deficiency that vitamin E couldn't cure, which Burr and his wife Mildred later identified as essential fatty acid (EFA) deficiency.
Observing that the annual per capita consumption of sugar in the United States had tripled over the preceding decades from 38 pounds to 115 pounds, Clarence Martin Jackson conducted a comprehensive analysis of the anatomy and tissues of rats fed Burr’s EFA-sufficient, 80% sucrose control diet and compared them to rats fed 45% sucrose or 45% corn starch (17). Neither the 45% sucrose diet nor the 45% starch diet produced fatty liver, but the 80% sucrose diet produced moderate to severe fatty liver with flattening and displacement of nuclei within the liver cells. Jackson warned that the liberal provision of cod liver oil, dried yeast and wheat germ satisfied the nutritional needs of the rats in all treatment groups, and that smaller amounts of sucrose may contribute to fatty liver in humans consuming nutritionally deficient diets.
This is rather remarkable, because much lower concentrations of sucrose started spontaneously producing fatty liver disease in lab rats in the late 1970s and early 1980s once the American Institute of Nutrition set standards for purified rodent diets that relied exclusively on isolated vitamins and minerals rather than whole-food supplements like cod liver oil, yeast, and wheat germ. We'll never know exactly how much choline was in Jackson's diet, but purified diets with 0.3% methionine and 0.8% choline, though to be adequate in these nutrients, produced moderate fatty liver when the sucrose concentration was increased from 20% to 25-35% (18).
Physicians and researchers had started pinning the blame on alcohol abuse for fatty liver back in the 1800s, so while research was first highlighting the role of sucrose in fatty liver, other research was doing the same for alcohol. In 1949, however, researchers showed that sucrose and ethanol had equal potential to cause fatty liver and the resulting inflammatory damage, and that increases in dietary protein, extra methionine, and extra choline could all completely protect against this effect (19).
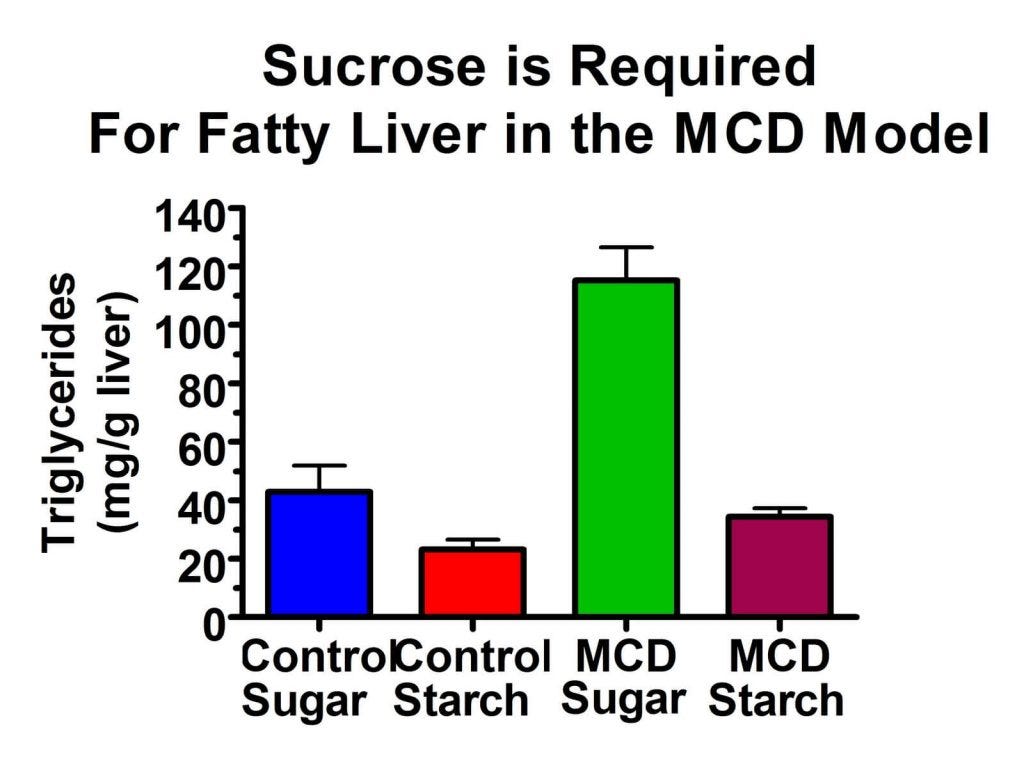
Conversely, much more recent research has shown that sucrose is a requirement for the development of fatty liver disease in a methionine- and choline-deficient (MCD) model. The MCD model of fatty liver disease is the oldest and most widely used dietary model. The MCD model produces not only the accumulation of liver fat, but massive inflammation similar to the worst forms of fatty liver disease seen in humans. What no one ever mentions about this diet is that it is primarily composed of sucrose and it's fat is composed entirely of corn oil!
As you can see here, the MCD diet resulted in only the most miniscule increase in liver fat when it was composed of starch (column 4 vs column 2), but when it was combined with sugar (green bar) it produced a massive increase in liver fat. The researchers also found that the MCD-sugar diet led to full-fledged inflammation, while the MCD-starch diet had no such effect (20). They found that on the sucrose diet, the fructose component led to the synthesis of fat in the liver, and because the choline level was deficient, the liver could not export that fat.
The picture that is clearly emerging from all of these studies is that fat, or anything from which fat is made in the liver, such as fructose and ethanol, are required for the development of fatty liver. But in addition to this some factor — overwhelmingly, it appears to be choline deficiency — must deprive the liver of its ability to export that fat.
Oxidative Stress May Impair Export of Liver Fat
An interesting test tube study using isolated cells found that incubating liver cells with PUFAs would suppress export of fats while incubating the liver cells with saturated fats, vitamin E, or a chelator of free iron would improve the export of fats. The results indicated that lipid peroxidation, that is, the oxidative destruction of PUFAs, causes liver cells to degrade their VLDL particles before they send them forth into what would be the bloodstream if the experiments were conducted in a live animal. They also provided preliminary evidence that this occurs in live animals by directly infusing their bloodstreams with DHA (21).
While it seems possible that excess PUFA might interact with iron overload, ethanol, or some other type of toxicity in order to impair the export of liver fat in the absence of choline deficiency, it nevertheless seems unlikely that excess PUFA in and of itself causes enough oxidative stress to make this happen, because even rats consuming 30% corn oil mentioned above fared fine if they were given enough choline. However, as we'll see below, excess PUFA are required for the inflammatory part of NAFLD. And, moreover, in humans there may be many interacting factors that “activate” the ability of excess dietary PUFA to suppress the export of fat from the liver.
MCD-Sugar Sets the Kindling, Corn Oil Lights the Fire
MCD formulas containing 20% fat as either lard or olive oil produce equivalent levels of NAFLD, suggesting that changes in the ratio of saturated to monounsaturated fats is not important (22). Substitution of carbohydrate, coconut oil, or beef tallow for corn oil similarly offers no protection against accumulation of fat in the liver nor on the injury to liver cells that causes increases in liver enzymes; all these substitutions, however, dramatically decrease lipid peroxidation and and the resulting inflammation (23). Corn oil probably promotes inflammation both by increasing vulnerability to lipid peroxidation because of its total PUFA content and by decreasing tissue levels of DHA because of its high omega-6-to-omega-3 ratio.
Choline is King!
I have to conclude from all these studies that choline deficiency plays a role in virtually every type of diet-induced fatty liver model. The fat has to be provided to the liver through either dietary fat or dietary lipogenic substrates like ethanol and fructose, and the fat has to be trapped by impaired export of fats from the liver. And choline deficiency seems to be the preeminent cause of this.
So does fructose cause fatty liver? Kind of. I'm not suggesting fructose is harmless or that you should go out an eat a bowl of fruit loops with your liver and eggs, but the loss of cholesterol-rich foods like egg yolks and organ meats as a result of cholesterol paranoia seems to be at the bottom NAFLD thus far.
There's just one question. Leptin deficiency and leptin resistance both cause fatty liver. And obese people are leptin resistant and the majority of obese people have fatty liver. Can fatty liver or choline deficiency cause leptin resistance? Or does leptin resistance cause cravings for choline-poor, fatty, and fructose-rich foods? Here's a web I'll try to untangle in future posts!
Join the Next Live Q&A
Have a question for me? Ask it at the next Q&A! Learn more here.
Subscribe
Subscribe or upgrade your subscription here.
Join the Masterpass
Masterpass members get access to premium content (preview the premium posts here), all my ebook guides for free (see the collection of ebook guides here), monthly live Q&A sessions (see when the next session is here), all my courses for free (see the collection here), and exclusive access to massive discounts (see the specific discounts available by clicking here). Upgrade your subscription to include Masterpass membership with this link.
Learn more about the Masterpass here.
Take a Look at the Store
At no extra cost to you, please consider buying products from one of my popular affiliates using these links: Paleovalley, Magic Spoon breakfast cereal, LMNT, Seeking Health, Ancestral Supplements. Find more affiliates here.
For $2.99, you can purchase The Vitamins and Minerals 101 Cliff Notes, a bullet point summary of all the most important things I’ve learned in over 15 years of studying nutrition science.
For $10, you can purchase The Food and Supplement Guide for the Coronavirus, my protocol for prevention and for what to do if you get sick.
For $15, you can pre-order a single format of my Vitamins and Minerals 101 book, my complete guide to nutrition, which I am currently working full-time on finishing.
For $25, you can pre-order a digital bundle of my Vitamins and Minerals 101 book.
For $29.99, you can purchase a copy of my ebook, Testing Nutritional Status: The Ultimate Cheat Sheet, my complete system for managing your nutritional status using dietary analysis, a survey of just under 200 signs and symptoms, and a comprehensive guide to proper interpretation of labwork.
For $35, you can pre-order a complete bundle of my Vitamins and Minerals 101 book.
For $250-$1499.99, you can work one-on-one with me.